New sensor mimics cell membrane functions

An MIT-led team designed a sensor that may be deployed to screen patients for hard-to-diagnose cancers, or metastatic tumors. The device draws inspiration from the membrane that surrounds all cells. Image: Courtesy of the researchers, edited by MIT News

“What we are aiming to do is develop the basic technology to enable a future portable device that we can integrate with cell phones and computers, so that you can do a test at home and quickly find out whether you should go to the doctor,” Qing says. Image: courtesy of the researchers.
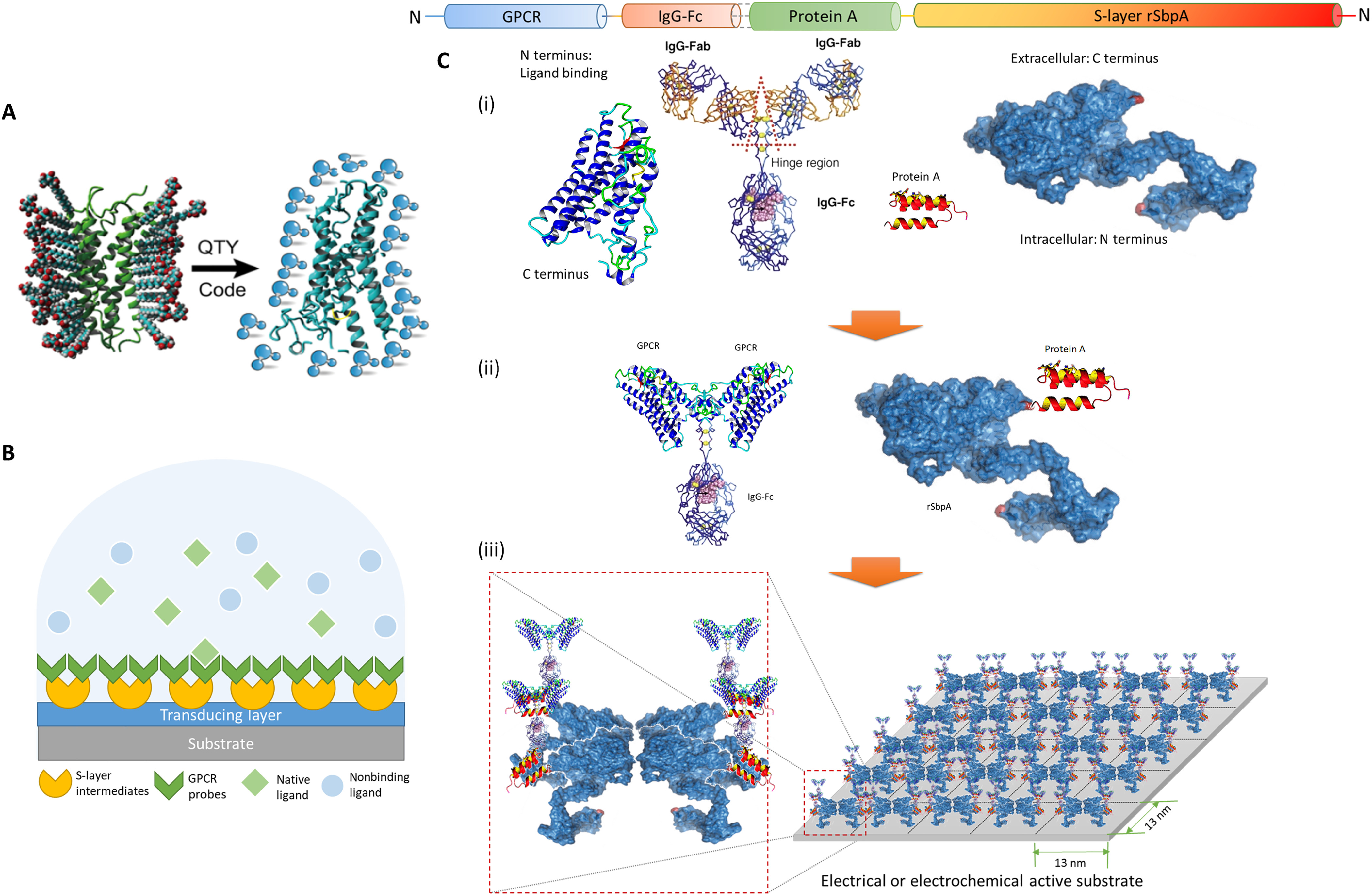
Schematic illustration of the architecture. (A) The QTY code transforms hydrophobic membrane receptors to water-soluble functional equivalents that do not require detergents to stabilize in aqueous solutions. (B) S-layer proteins and solubilized receptors are sequentially coated onto designated sensing devices. Target analytes are specifically recognized in aqueous media, while the detection signal is transduced for electrical responses. GPCR, G protein–coupled receptor. (C) Sequence layout of the dual-monolayer construct. (i) The four elements of the construct: redesigned water-soluble membrane receptor, heavy chain Fc region of immunoglobulin G (IgG) protein, Fc-binding fragment of protein A, and two-dimensional (2D) crystal-forming S-layer protein. (ii) Solubilized receptors are fused to IgG-Fc to serve as the biospecific probes, while S-layer proteins are fused with protein A fragments to connect probes to the device. (iii) Sequentially, S-layer/protein A fusion proteins reproduce its native nanoscale 2D crystalline lattice on the substrate, after which the receptor probes with Fc fusions were applied to bind the intermediate layer in high affinity and capacity. The specific construct presented here (rSbpA-ZZ/CXCR4QTY-Fc) has p4 symmetry, a lattice parameter of ~13 nm2, and a receptor density in the order of ~1012 U/cm2. Image: Rui Qing, et al., Scalable biomimetic sensing system with membrane receptor dual-monolayer probe and graphene transistor arrays.Sci. Adv. 9, eadf1402 (2023).

